 The geometry and shape of ethane will be the same owing to the absence of lone pair of electrons. In ethane, carbon is a central atom and it has no lone pair of electrons. The molecular geometry of a compound is determined by valance shell electron pair repulsion (VSEPR) theory.Īccording to this theory, the shape and geometry of the molecule depend on the number of bonding electrons and lone pair of electrons. Now, let us move towards from 2D representation to 3D representation of the molecule i.e., molecular geometry of ethane. Therefore, there is no lone pair of electrons on any atom of ethane. If we observe, we can see that all 14 valance electron has been used for bond formation. Every hydrogen atom is surrounded by two electrons, leading to duplet formation. The total number of electrons around each carbon atom is 8 electrons and hence, it has completed its octet. We can count the total number of valance electrons in the Lewis structure of ethane, which is equal to 14. Therefore, it forms 4 bonds, one with neighboring carbon atoms and three with three hydrogen atoms by sharing electrons. In ethane, we have two carbon atoms and 6 hydrogen atoms and hence, the total number of valence electron are (2 X 4) + (1 X 6) = 14.Ī carbon atom has 4 valance electrons and it needs 4 more electrons to complete its octet. The valence electron for carbon (1s22s22p2) and hydrogen (1s1) is 4 and 1, respectively. Step 1: Determining the total number of valence electrons in the molecule. Let us draw a Lewis structure of ethane step by step. every atom tends to complete its octet ( 8 electrons) either by gaining or losing electrons except Hydrogen and Helium as they complete their duplet. Lewis structure is a 2D representation of the compound, which represents only the valance shell electrons of the atoms in the molecule. Let us discuss the basic concepts of ethane such as its Lewis structure, polarity, hybridization of carbon atom in ethane, and its Molecular orbital (MO) diagram to understand its chemical bonding in terms of molecular orbitals. Here are some methods of preparation of ethane:ĬH3 − CH2Cl + H2 - Zn/H+ -–> CH3 − CH3 + HClĢCH3Cl + 2Na -Dry ether-> CH3 − CH3 + 2 NaClĢ CH3COONa + 2H2O -–Electrolysis-> CH3 − CH3 + 2NaOH + H2 + 2CO2 It can also be prepared from ethene, ethyl chloride, and sodium acetate in the laboratory. Ethane is obtained from natural gas and petroleum industrially. The flashpoint of ethane is -135 ☌ and hence, its vapors ignite easily by an ignition source. The melting and boiling point of ethane are -182.8 ☌ and -89 ☌, respectively.  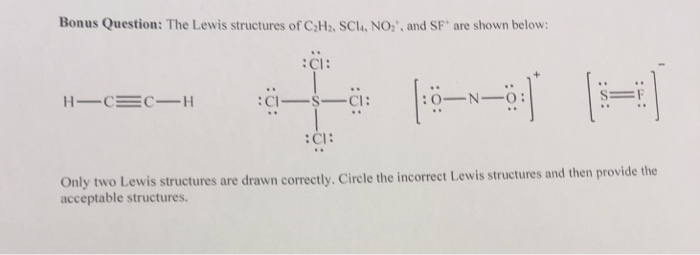 
Ethane can also be written as CH3-CH3.Įthane is colorless and odorless gas at standard temperature and pressure. Saturated hydrocarbons are further classified into alkane (open chain of carbon atoms) and cycloalkane (closed chain of carbon atoms). Saturated hydrocarbons are those hydrocarbons, which contain carbon-hydrogen and carbon-carbon single bonds. Hydrocarbon is an organic compound, which contains only carbon and hydrogen. C2H6, known as ethane, is a saturated open-chain hydrocarbon or we can say that it comes under the alkane family. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
